Almost two million government-approved antibody tests imported into Australia and sold to GPs,hospitals and aged care clinics are so inaccurate they should not be used to diagnose COVID-19.
Testing at the Doherty Institute this week has found the tests,designed to tell if a person has already been infected by and recovered from coronavirus,are about as accurate at picking up COVID-19 antibodies as flipping a coin.
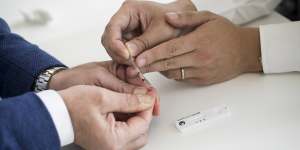
The government's fingerprick tests have been found not to work well enough.Dominic Lorrimer
The Sydney Morning Heraldand The Age that the federal government has received official advice that the 1 million antibody tests it purchased are not fit for purpose. The government has repeatedly refused to say how much it paid for the tests,or who they were purchased from.
The government also tried to purchase 500,000 tests manufactured by Chinese company Wondfo – the same company that supplied tests to the UK government which are now embroiled in scandal.
Wondfo denied problems with its tests following a report by theNew York Timesthat the UK government had concluded they did not work.
The Wondfo test,and the two analysed by the Doherty Institute,are among 32 antibody tests approved for use in Australia by the Therapeutic Goods Administration. The Wondfo test was approved three times – the last approval coming on April 17,a day after theNew York Times’report.
Unusually,the regulator did not do its own independent studies to check if those tests were accurate before approving them.